Mechanical Imbalance Between Normal and Cancer Cells Drives Epithelial Defense Against Cancer
In collaboration with the group of Prof. Tamal Das
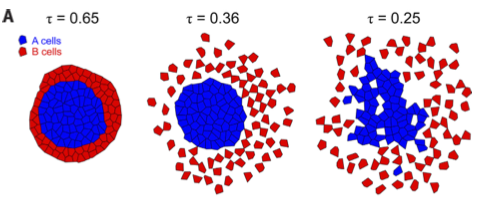
Cell competition in epithelial tissues helps maintain homeostasis by eliminating transformed cells that express cancer-promoting oncoproteins. Although this process is known to involve mechanical forces, the specifics were unclear. Our research used advanced stress measurement techniques and theoretical modeling to reveal a mechanical imbalance between normal and transformed cells that drives this competition. We found that transformed cells experience compressive stress, making them more compact during competition. This stress arises from their higher collective compressibility compared to normal cells, due to weaker cell-cell adhesions and reduced E-cadherin. These findings provide new insights into how epithelial tissues defend against cancer by mechanically targeting abnormal cells.

- Gupta, P., Kayal, S., Tanimura, N., Pothapragada, S.P., Senapati, H.K., Devendran, P., Fujita, Y., Bi, D., & Das, T. “Mechanical imbalance between normal and transformed cells drives epithelial homeostasis through cell competition.” eLife (2025). DOI